(Asian independent) September 16 is celebrated across the globe as World Ozone Day and it is officially known as the International Day for the Preservation of the Ozone Layer. This day was established by the United Nations in 1994 and this day marks the anniversary of the signing of the Montreal Protocol in 1987, a landmark international treaty designed to phase out substances responsible for ozone layer depletion. World Ozone Day serves as a reminder call to renewed action in the face of continuing threats being faced by the ozone layer. It highlights the critical role the ozone layer plays in protecting life on Earth and underscores the collective responsibility of humanity to preserve this vital atmospheric shield.
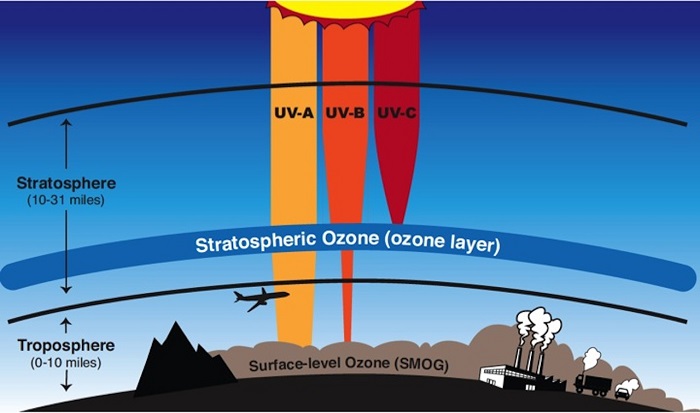
Formation of the Ozone Layer
Ozone (O₃) is a triatomic molecule consisting of three oxygen atoms. It is formed in the stratosphere, approximately 15 to 35 kilometers above Earth’s surface, through a process driven by solar ultraviolet radiation. The formation of ozone follows the Chapman Cycle, first proposed by British scientist Sydney Chapman in 1930:
- Oxygen Photodissociation:
Ultraviolet light (wavelength < 240 nm) splits molecular oxygen (O₂) into two atomic oxygen (O): - Ozone Formation:
The atomic oxygen (O) reacts with molecular oxygen (O₂) in the presence of a third molecule (M, often N₂ or O₂) to form ozone (O₃): - Ozone Photolysis:
Ozone molecules absorb UV-B radiation (280–315 nm) and dissociate back into molecular and atomic oxygen:
This dynamic equilibrium ensures a relatively stable ozone concentration, forming what is globally recognized as the ozone layer.
Importance of the Ozone Layer
The primary function of the ozone layer is to shield life on Earth from the sun’s harmful ultraviolet radiation. Specifically, it absorbs the majority of UV-B and nearly all UV-C radiation, both of which are highly harmful to the biological systems. Without the protection of ozone layer humanity can be effected in the following ways:
- Increased UV radiation would lead to a surge in skin cancers, cataracts and immunological deficiencies in humans.
- Agricultural productivity would decline as many crops are sensitive to UV exposure.
- Marine ecosystems, especially phytoplankton populations forming the base of the aquatic food web, would face severe disruption.
- Synthetic polymers and materials would degrade faster, leading to economic losses.
In essence, the ozone layer functions as Earth’s natural sunscreen making it crucial for ecological and human health.
Causes of Ozone Depletion
The discovery of the ozone hole over Antarctica in the 1980s brought widespread attention to the anthropogenic causes of ozone layer depletion. Chief among these are chlorofluorocarbons (CFCs), halons and other ozone-depleting substances (ODS) which, though once widely used in refrigeration, air conditioning, foam production and aerosol propellants are highly stable compounds that persist in the atmosphere for decades.
When these chemicals reach the stratosphere, ultraviolet radiation breaks them down releasing chlorine (Cl) and bromine (Br) atoms. These atoms catalytically destroy ozone through the following reactions:
- Chlorine cycle:
One chlorine atom can destroy thousands of ozone molecules before being deactivated.
Factors Exacerbating Ozone Depletion
Several environmental factors exacerbate ozone depletion, notably:
- Polar Stratospheric Clouds (PSCs):
Formed under extreme cold conditions in polar regions, PSCs provide surfaces for chemical reactions that convert stable chlorine compounds into active chlorine, accelerating ozone breakdown. - Climate Change:
Rising greenhouse gas concentrations affects the stratospheric temperatures, altering ozone chemistry and circulation patterns potentially exacerbating localized depletion. - Volcanic Eruptions:
Massive volcanic eruptions could inject particles into the stratosphere, increasing PSC formation and promoting chlorine activation.
Steps for Ozone Layer Conservation
In response to global alarm, the Montreal Protocol on Substances that deplete the Ozone Layer (1987) was established, representing a landmark in international environmental policy. The protocol and its amendments have led to:
- A phased reduction and eventual ban of CFCs, halons and related compounds.
- Promotion of alternative, ozone-friendly substances such as hydrofluorocarbons (HFCs).
Beyond policy frameworks, essential conservation steps include:
- Monitoring and Research:
Continuous satellite-based and ground-based monitoring of ozone levels and stratospheric conditions. - Public Awareness and Industry Compliance:Educating stakeholders about the environmental impact of ozone depleting substances ODS and promoting industrial transitions to eco-friendly technologies.
- Scientific Innovation:
Development of sustainable refrigeration and manufacturing alternatives.
The Need for International Joint Efforts
Ozone depletion is an international problem that transcends national borders. The Montreal Protocol remains the most successful environmental treaty demonstrating how international cooperation can lead to measurable environmental recovery. Nonetheless, continued vigilance is essential:
- Strengthening global compliance and enforcement.
- Supporting developing nations in transitioning away from ozone depleting substances ODS.
- Addressing new challenges, such as the environmental impact of HFCs, which, while not harmful to ozone are potent greenhouse gases contributing to climate change.
Joint global action is imperative to prevent the reversal of gains and ensure a fully recovered ozone layer by mid-21st century, as projected by current scientific models.
World Ozone Day serves as an important reminder of humanity’s capacity to tackle environmental crises through science, international policies and global unity. The ozone layer remains an invisible but critical guardian of life protecting ecosystems and human health from harmful ultraviolet radiation. Continued scientific research, international collaboration and sustainable technological innovation must remain at the forefront of the global efforts. The future of the planet’s habitability depends on how diligently humanity care for this fragile atmospheric shield today.
SURINDERPAL SINGH
FACULTY IN SCIENCE DEPARTMENT
SRI AMRITSAR SAHIB PUNJAB.